Antiproliferative/Antimetabolic
Agents
Anti-Alpha
4 Integrin Inhibitors
Biology

Classes of immunosuppression
- Glucocorticoids
- mTOR
inhibitors
a.
Sirolimus
- Calcineurin
inhibitors
- Cyclosporin
- Tacrolimus
- Antiproliferative/antimetabolic
agents
- Azathioprine
- Mycophenolate
- Biologics
- TNF
inhibitors
i. Entanercept
ii. Adalimumab
iii. Golimumab
iv. Certolizumab
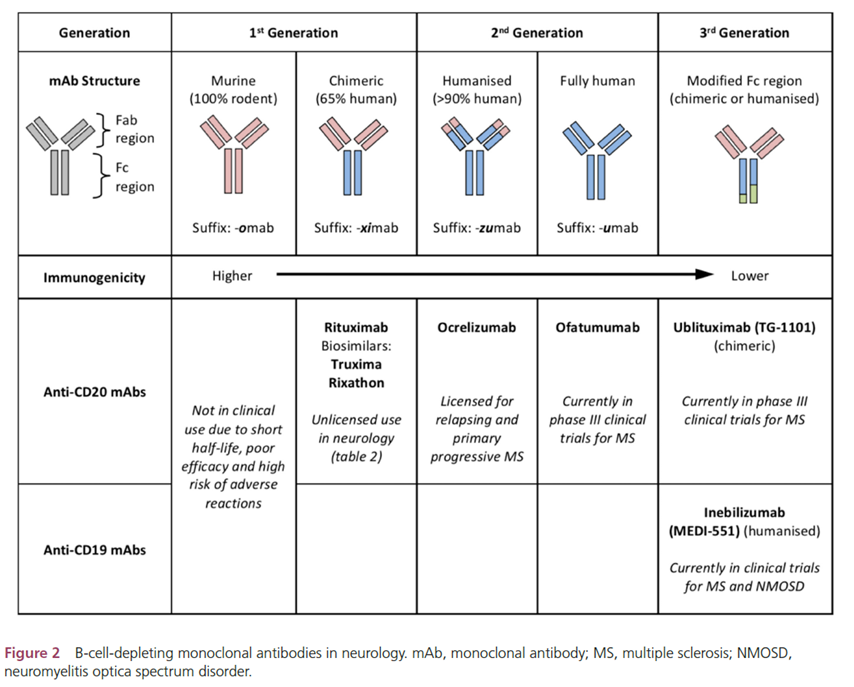
- Anti-CD-20
i. Rituximab
ii. Ocrelizumab
Risk management – Infection
Consider testing for infection and immunisation status:
|
All patients |
|
|
Hepatitis
B (Full panel) |
Hep BsAg (Infection – contraindication to treatment) Hep BsAb (?Immune) Hep BcAb (latent infection – seek advice if positive, may need prophylaxis) |
|
Hepatitis
C |
|
|
HIV |
|
|
Varicella |
VZV IgG OR Documented history of vaccination (Patients who have been vaccinated do not necessary show positive IgG response, but still have protection) |
|
Tuberculosis |
Quantiferon assay Consider CXR in high risk individual |
|
Consider |
|
|
EBV, CMV,
HSV |
|
|
Syphilis |
|
|
Strongyloidies |
|
|
JCV |
|
Vaccination
Consider Vaccination for:
|
Vaccination |
Who to consider |
Comments |
|
Hepatitis B |
At risk: -
Family contacts, ATSI, migrant communities,
IVDU, Prisoners, Health care workers/Police force, MSM, |
Standard is 3 vaccinations – 0, 1 and 4 months Accelerated vaccination over 1 month can be considered – limited evidence and likely reduced efficacy. |
|
Varicella |
Test VZV serology or document vaccination history - If VZV negative and no documented history of vaccination -
Older patients with positive VZV serology -
>70yrs, however consider for patients >50 years if going on
immunosuppression. |
– Varicella vaccine – ideally 2 doses 1 month apart, however even one dose has reasonable efficacy. - Zostavax can be considered. Given as single dose. NOTE: Live vaccines – thus must be given at least 4 weeks prior to starting immunosuppression. |
|
Influenza |
All patients |
Annual |
|
Pneumococcal |
>70 years (regardless of other risk factors) ATSI >50years Any patient with risk factors (previous pneumococcal infection, chronic disease, asplenia). Immunosuppressive medication is not listed as specific risk factor. |
Single dose of 13PCV Dose of 13 PCV and later 23 PCV Dose of 13 PCV and later 23 PCV |
Avoid Live vaccines:
• Immunosuppression should be delayed until 4-6 weeks after live vaccination
|
Live
attenuated parenteral vaccines |
Live
attenuated oral vaccines |
||
|
Viral |
Bacterial |
Viral |
Bacterial |
|
Japanese encephalitis (Imojev) Measles-mumps-rubella
(MMR) |
BCG |
Oral rotavirus vaccine |
Oral typhoid vaccine |
Prophylaxis:
Bactrim in non-transplant setting
Steroid treatment
• Should be considered for patients on >/= 20mg/day for >1month
• This recommendation is far more important if co-existent risk factors exist:
o Malignancy
o Respiratory disease
o Co-administration of other immunosuppressant’s.
Other immunosuppression
• Azathioprine – consider if >3mg/kg/day
• Methotrexate – consider if >0.4mg/kg/week
In heavily immunosuppressed patients (e.g. post transplant)
• Bactrim
o Protects against – PCP, Toxoplasmosis, Isospora, norcardia, listeria and others
o Give for at least 3 months to lifelong
o Alternatives to bactrim include dapsone or atovaquone
• Anti herpes virus prophylaxis
o Usually valganciclovir
o Protects against CMV primarily (also HSV, VZV, EBV)
o By preventing viral infection-
- Reduces incidence of secondary fungal infections
- Reduces incidence of PTLD
- May reduce BOOP in lungs and vasculopathy in heart Tx
o 3-6 months often used
o Some units do not use routinely but monitor carefully and treat ASAP
• HBV prophylaxis
o If core positive but surface antigen negative then prophylaxis warranted
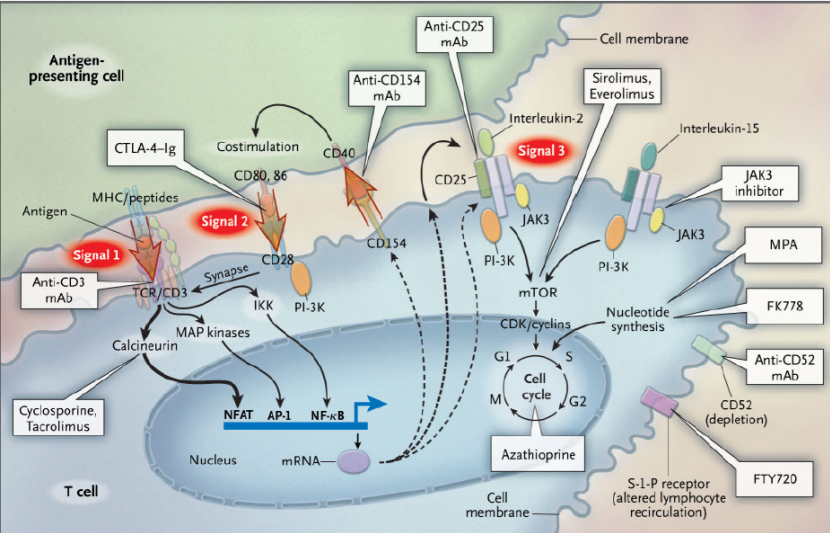
Calcineurin
Inhibitors
· Bind to
intracellular proteins and subsequently inhibit calcineurin
· This prevents
(calcineurin from) activating transcription factor NFAT
· The primary effect
of this is reduced production of IL-2
· Other cytokines
down regulated:
o
TNF-a, IL-3, IL-4, IFN-gamma
· Also reduces
transcription factors NF-kB and AP-1
· Action primarily
effects helper T-cells
· Does not have myelosupressive effect
· Reduces activation
of immune system, has little effect once it is activated.
Cyclosporine
·
Binds cyclophilin and the complex binds calcineurin
and inhibits its effect on transcription
·
Metabolised by CYP3A4
o
Multiple drug interactions
Toxicity
·
Nephrotoxicity
o
Acute reversible
o
Chronic irreversible
§ (Obliterative
arteriolopathy – suggests endothelial damage)
·
HTN
·
Gingival hyperplasia (?can
be Rx with metronidazole)
·
Hirsutism
·
Neurological – Tremor common (usually mild),
encephalopathy and headache
·
Induction of HUSTTP
·
DM induction (and increased insulin requirements)
·
Dyslipidaemia
·
Increase K+, low Mg2+
·
GI upset (common, 50%, but not limiting)
·
Increased malignancies (more due to the immunosupression in general)
Tacrolimus
o
Also binds calcineurin in a complex (with FKBP12)
o
Similar efficacy to cyclosporine at std. doses
Toxicity
o
Similar to cyclosporine:
o
Nephrotoxicity
o
HUSTTP
o
Less likely to cause other cyclosporine SE (HTN,
hyperlipidaemia, gingival hyperplasia, hirsuitism)
o
More likely to induce DM
o
Neurotoxicity more likely
o
GI upset more common
mTOR
inhibitors
Sirolimus and Everolimus
Use
·
Transplant – heart and renal
·
Treatment of Renal cell carcinoma
Interactions
·
Metabolised via P-450
·
Numerous interactions – caution when changing dose
·
Interaction with cyclosporine
·
Everolimus trough levels can
be monitored
Toxicity
·
Marrow suppression
o
Anaemia (30-60%)
o
Thrombocytopaenia
o
Leukopenia
·
Metabolic
o
Hyperlipidaemia
§ TG and cholesterol
elevated (via inhibition of LPL)
o
DM – prolonged use can induce DM
·
HUSTTP
o
Has occurred in rare cases usually when
cyclosporine co-administered.
·
Renal function
o
Minimal impact on its own, may be synergistic
effect with cyclosporine
·
Respiratory
o
Associated with interstitial lung disease
·
Impaired wound healing
·
Antiproliferative/Antimetabolic
Agents
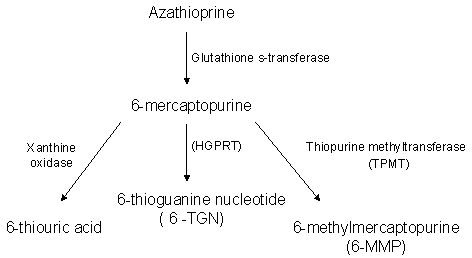
Azathioprine
- Converted to
6-mercaptopurine
- Then
converted to different metabolites
- 6-TGN –
induces immune cell apoptosis
- Interferes
with purine metabolism
- Excess
converted to toxic metabolite (6MMP)- responsible for
hepatotoxicity
- TPMT enzyme
differences can determine effects
- 10% of
population are slow metabolises
- <1% have
no enzyme and are at risk of severe toxicity – especially bone marrow
toxicity
Toxicity/SE
• Myelosuppression (~27%)
o Leukocytopenia >thrombocytopenia>anaemia
• GI upset (mainly upper) – occurs in 25%
• Hepatitis (rare, usually mild elevation of LFTs in 5%)
• Pancreatitis
• Infection (particularly if leukocytopenia)
• Neoplasm/lymphoma
• Pregnancy – probably safe to use, some SE however NOT teratogenic
• Breastfeeding – probably safe
Interactions
• Allopurinol – blocks xanthine oxidase which metabolises azathioprine
o Must decrease dose by 25-33% or avoid combination
• Co-administration of ACE-I may increase risk of leukopenia
Dose:
• Start: 0.5-1.0mg/kg/day
• Increase over 3-4 weeks to target 2.0-2.5mg/kg/day
• Caution at extremes of body weight – may be better guided by blood testing.
• Lower dose if combination with allopurinol
• Lower dose if slow metaboliser (based on TPMT activity)
Monitoring
• FBC/LFT
o Every 2 weeks when starting (or changing dose) – continue for 6 weeks once stable dose reached.
o Then - Monthly for 3 months
o Then –3 monthly thereafter.
o If lymphocyte count <0.5 – consider dose reduction
o If total WCC <1.5 – consider withholding treatment
• MCV
o Dose related increase
o Roughly correlates with efficacy
o Not accurate enough to use as definitive marker of action – better used as marker of compliance.
• 6-TG and 6-MMP levels
o 6TG
- >400 correlates with myelosuppression
- 235-450 correlates with clinical efficacy
- <235 Poor efficacy
o 6MMP
- >5700 correlates with hepatotoxicity
o Check at:
- 4 weeks after starting or changing doses
- Then ~3 months after starting
- Then Annually
o Results:
- See table:
|
|
|
6-MMP |
|
|
|
|
Low |
High |
|
6-TG |
Low |
Subtherapeutic
dosing (or poor compliance) |
Hypermethylation
occurring Split
or reduce dose and add allopurinol |
|
High |
Supratherapeutic
effect – reduce dose |
Supratherapeutic
effect – reduce dose |
|
• Malignancy monitoring
• Advice to monitor for infections (and education regarding vaccination)
• Advice regarding pregnancy
Mycophenolate mofetil (MMF)
- Metabolises
to active agent – Mycophenolic acid (MPA)
- Inhibitor of
IMP dehydrogenase (IMPDH) – an important enzyme in guanine (purine)
synthesis
- Lymphocytes are
dependent on de-novo synthesis while other cells use salvage pathway –
hence selective effect.
Uses
·
Transplantation
·
SLE – renal crisis (as alternative to
cyclophosphamide)
Pharmacokinetics
·
Metabolised by glucuronidation, mainly excreted in
urine
·
Watch dose in renal failure
Toxicity
- Leukopenia
- GI upset –
Diarrhoea, Vomiting, dyspepsia, nausea, abdominal cramping (75%)
- ?Increased risk of infections
- CMV
- May be
protective against PCP
·
Raised LFTs (25%)
·
Pregnancy - Not safe, known teratogenic
Dose
·
Mycophenolate mofetil 1-1.5g bd
·
Mycophenolate sodium 720mg bd
·
If GI side effects can try:
o
Give with food
o
Give tds or qid
o
Switch to mycophenolate sodium
o
Reduce dose
Monitoring
·
FBC weekly for 1 month then:
·
FBC every 2 weeks for 2 months then:
·
FBC every 1-3 months ongoing
·
Levels can be done but are complicated (see AMH)
Leflunomide (Arava)
- Inhibits
di-hydro orotate dehydrogenase (DHODH) resulting in inhibition of the
pyrimidine rUMP
- Inhibits
- leukocytes
adhesion, Memory T cells, Dendritic cells
- NF-KB
- Various
other proposed mechanisms
- May have
anti-BK virus effect
- No major
modification needed for renal or hepatic dysfunction
Indications
·
Rheumatoid arthritis – often in combination with
MTX in patients who have failed monotherapy
·
BK virus infection in renal transplant patients
(experimental)
Adverse
o
HTN
o
GI upset
o
Hepatotoxicity
- ~10%,
usually reversible
o
Myelosupression
- Leukopenia
is very rare
o
Not safe in pregnancy
Methotrexate
- DHFR
inhibitor
- This does
not seem to account for its entire effect and alternative mechanism
affecting adenosine signalling is also proposed
Pharmacokinetics
- 80-90%
excreted unchanged in urine – therefore need to dose adjust in renal
impairment
- Oral, SC and
IM dosing (later may be used due to variable bioavailability)
·
Usually given weekly – minimises SE without effect
on efficacy
·
Concurrent folic acid – 1mg daily, may reduce SE
and has not been demonstrated to reduce efficacy. Weekly folinic acid
(leucovorin) may be similarly used.
·
Drug interactions
o
Caution using concurrent high dose bactrim (which also inhibits DHFR)
o
Leflunomide increases risk of raised LFTs
Toxicity
·
Depends on dose used (high doses used in
chemotherapy)
Serious
·
Hepatotoxicity
o
Transaminitis in 10%, more severe disease rare
o
AST level most predictive of damage
o
Consider pause or cessation if > x2 ULN
o
Consider reduced dose if ongoing mild elevation
·
Pulmonary
o
Many different types but hypersensitivity
pneumonitis is most common
o
Can occur at any dose
o
Acute or chronic
o
Restrictive RFTs on reduction
o
Improve with cessation
o
May be eosinophilia
·
Myelosupression
o
Neutropaenia
§ Dose dependent
§ Stop if count <1.0
o
Lymphopaenia
§ Mild reduction
common
§ Consider reduction
if <0.8
o
Pancytopaenia
§ Higher risk in
elderly and renal impairment or using Bactrim
§ Cease MTX and
start high dose folinic acid (15mg QID IV)
·
Nephrotoxicity
o
Precipitation of MTX crystals and tubular injury
·
Pregnancy – absolute CI
o
Cease for 12 weeks prior in male and female
·
Malignancy
o
Lymphoma and leukaemia risk is double, cautation unclear
o
Probable increase in non-melanoma skin cancer
·
Infection risk
o
No general increase in risk
o
Shingles risk is increased
o
PJP rare case reports
Common
·
GI upset
o
Nausea and vomiting and less commonly diarrhoea
o
Usually up to 72 hours post dose
o
Try nocte dose or increase folic acid up to 5mg 6
days/week, or change to folinic acid 7.5mg 12 hours
post dose
·
Stomatitis and oral mucosal lesions
·
Macular rash
·
Alopecia
·
CNS – headache, fatigue
o
Often in 72 hours post dose
o
Try reduction of dose by 5mg if severe
·
Alopecia
·
Fever
·
Macrocytosis
Cyclophosphamide
·
Alkylating agent
·
Severe life or organ threatening disease
·
Converted in liver to a variety of metabolites
·
Excreted in urine – need to renal adjust
·
Oral is more effective in many cases but has higher
toxicity than IV
Toxicity
·
Lymphopenia
o
Almost universal with higher doses
o
Concurrent steroids may worsen this
o
Should reduce therapy if WCC < 3 to 4.
·
Bladder toxicity
o
Haemorrhagic Cystitis - Acrolein – is metabolite
which is toxic to the bladder
§ Superhydration – fluid and
frusemide to achieve UO >150ml/hr is effective
§ MESNA binds
acrolein and can be given to reduce toxicity
§ Treatment is difficult -
hydration, bladder washout, may need instillation of formalin or alum to stop
bleeding, surgical options
o
Bladder cancer risk is increased and yearly urine
screening for haematuria is recommended.
o
Cumulative dose and previous haemorrhagic cystitis
are risk factors for TCC development
o
It is thought that BK virus infection has a role in
inducing toxicity
·
Infertility
o
Female
§ Relates to
increased cumulative dose, oral dose and age (>25yrs), risk of complete
ovarian failure in low risk groups 10-50%.
o
Male
§ Risk less clear ?>50%
o
Use of GnRH agonists to induce gonal
quiescence may be of benefit
o
Preservation of ovum and sperm
·
Teratogenic
·
Risk of infection increased – of multiple organisms
o
PCP prophylaxis is recommended
o
VZV reactivation
·
Malignancy
o
Increased risk of lymphoma
o
Increased risk of bladder cancer
Biologics
|
TNF inhibitors |
|
|
|
|
Infliximab |
|
|
|
|
Entanercept |
|
|
|
|
Adalimumab |
|
|
|
|
Golimumab |
|
|
|
|
Certolizumab |
|
|
|
|
|
|
|
|
|
IL-1 Inhibition |
|
|
|
|
Anakinra |
|
|
|
|
IL-2 Inhibitors |
|
|
|
|
Basiliximab (Simulect) |
|
|
|
|
Daclizumab
(Zenapax) |
|
|
|
|
Alpha-4-Integrin AB |
|
|
|
|
Natalizumab
(Tysabri) |
|
|
|
|
|
|
|
|
|
|
|
|
|
TNF inhibitors
Infliximab
(Remicade)
- Chimeric
(human-mouse) IgG, TNF-a monoclonal antibody
- Indications
- RA
- Ankylosing
spondylitis
- Psoriatic
arthritis
- Psoriasis
- Crohn’s
- UC
Entanercept
(Enbrel)
- recombinant
soluble p75 TNF receptor :Fc fusion protein (also
targets TNF-beta)
- There are two
naturally occurring TNF receptors – the ‘active’ one on cell surfaces and a
soluble ‘decoy’ one produced to decrease TNF action.
- This drug
utilises the later, soluble TNF receptor in a more stable, long lasting
form which clears the TNF.
- Indications
- RA
- Anklyosing spondylitis
- Psoriatic
arthritis
- Chronic plaque
psoriasis
Adalimumab
(Humira)
- Recombinant
humanised monocloncal TNF-a AB
- Indications
- Adult RA
- Psoriatic
arthritis
Golimumab
(Simponi)
- As per
Adalimumab
Certolizumab
pegol (Cimzia)
- As
per Adalimumab but pegylated
Adverse
effects of TNF inhibitors
- Infection
- Opportunistic
infections
- Serious
bacterial infections (high dose infliximab)
- Reactivation
of latent TB (infliximab >others)
- Pneumococcus/influenza
vaccine should be given before commencement
- Administration
- Injection
site reactions
- Infusion
reactions
- Hypersensitivity
- Serum-sickness
like reactions
- Malignancy
- Lymphoma
(2-6x)
- Controversial
- Haematological
- Pancytopenia,
aplastic anaemia
- CVS
- Increased
risk of CHF and mortality at high dose infliximab
- Autoimmune
- Human anti-chimeric
antibodies to infliximab
i. Reduced incidence
with MTX/AZA
ii. Abrogates use of
infliximab if it occurs
- Drug-induced
lupus
- Development
of autoantibodies
- Neurological
- De novo
demyelinating disease
- Parkinsons
disease
IL-1 Inhibition
Anakinra (Kineret)
- Recombinant,
IL-1Ra (competitive ihibitor of IL-1 binding
IL-1R)
- Indications
- Active RA
(combined with MTX but not TNF-Inhibitors)
IL-2 inhibition
- Basiliximab (Simulect)
- Daclizumab (Zenapax)
- Antagonize
IL-2R
- Used for
renal transplantation
- Ongoing
trials in other conditions
Anti-Alpha 4 Integrin Inhibitors
- Natalizumab
(Tysabri)
- Inhibits
T-cell entry into CNS
- Used in MS
Anti-CD20 Antibodies
Rituximab
·
RA - Proven useful in patients with rheumatoid
arthritis
o
Role of pathological antibodies/immune complexes in
disease.
o
Usually used in combination with methotrexate, NOT
used in combination with anti-TNF therapy
·
SLE – has been used successfully
Adverse effects
·
Infusion reactions
o
30-45% at first infusion, ?due
to release of cytokines from destroyed cells
o
Hypotension, throat tightness, rash, nausea, back
pain
o
Premedication may help
·
Infection
o
Overall not increased relative to patients on
methotrexate
o
Maybe a small increase in Pneumocystis pneumonia
·
PML - May be small increased risk
·
Impaired vaccination response
·
Human anti-chimeric antibody – up to 5%
o
Decrease efficacy
o
Increased risk of infusion reactions
·
Monitoring
From Practical Neurology



Summary of Side Effects
|
Cyclosporine |
Nephrotoxicity HTN Gingival
hyperplasia Hirsutism Neurological
– Tremor common Induction
of HUSTTP DM
induction Dyslipidaemia Increase
K+, low Mg2+ GI
upset (common, 50%, but not limiting) |
|
Tacrolimus |
As for
cyclosporine with the following exceptions: More
DM More
neurological SE More
GI upset No
gingival hyperplasia or hirsuitism |
|
Azathioprine |
|
|
Mycophenolate |
Leukopenia GI -
Diarrhoea, dyspepsia, nausea, abdominal cramping (75%) ?Increased risk of infections (May be protective against
PCP) Raised
LFTs (25%) Pregnancy
- Not safe, known to be teratogenic |
|
Sirolimus |
|
|
|
|
|
|
|
|
|
|
|
|
|